Products
PIRIMIPHOS-METHYL
Insecticide, acaricide
organophosphorus
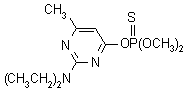
NOMENCLATURE
Common name pirimiphos-methyl (BSI, E-ISO, ANSI, ESA);
pyrimiphos-méthyl ((m) F-ISO)
IUPAC name O-2-diethylamino-6-methylpyrimidin-4-yl O,O-dimethyl
phosphorothioate
Chemical Abstracts name O-[2-(diethylamino)-6-methyl-4-pyrimidinyl] O,O-dimethyl
phosphorothioate
CAS RN [29232-93-7] EEC no. 249-528-5 Development
codes PP511 (ICI) Official codes OMS
1424
PHYSICAL CHEMISTRY
Composition Tech. material is 88% pure. Mol.
wt. 305.3 M.f. C11H20N3O3PS Form Straw-coloured
liquid. M.p. 15-18 ºC (tech.) B.p. Decomposes
on distillation. V.p. 2 mPa (20 ºC); 6.9
mPa (30 ºC); 22 mPa (40 ºC) KOW logP
= 4.2 (20 ºC, unionised) Henry 6 ´ 10-2 Pa
m3 mol-1 (calc.) S.g./density 1.17
(20 ºC); 1.157 (30 ºC) Solubility In
water 11 (pH 5), 10 (pH 7), 9.7 (pH 9) (all in mg/l, 20 ºC). Miscible
with most organic solvents, e.g. alcohols, ketones, halogenated hydrocarbons. Stability Hydrolysed
by concentrated acids and alkalis; DT50 2-117 d (pH range 4-9,
most stable at pH 7). In sunlight, aqueous solution had DT50 <1
h. pKa 4.30 F.p. >46 ºC
APPLICATIONS
Biochemistry Cholinesterase inhibitor. Mode
of action Broad-spectrum insecticide and acaricide with contact
and respiratory action. Penetrates the leaf tissue and exhibits translaminar
action. Uses Control of a wide range of insects
and mites in warehouses, stored grain, animal houses, domestic and
industrial premises; chewing insects, sucking insects, boring insects,
and mites on vegetables, ornamentals, bulb flowers, sugar cane, maize,
sorghum, rice, citrus and other fruit, olives, vines, alfalfa, cereals,
etc.; and glasshouse pests (especially whitefly, thrips, mealybugs,
aphids, and mites) on tomatoes, cucumbers, capsicums, aubergines, and
other glasshouse crops. Formulation types AE;
DP; EC; FU; HN; KN; LS; SG; UL. Compatibility Miscible
with common insecticides and fungicides. Mixing with strongly alkaline
or acidic substances should be avoided.
