Products
CLOPYRALID
Herbicide:; pyridinecarboxylic acid
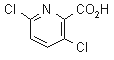
NOMENCLATURE
clopyralid
Common name clopyralid (BSI, ANSI, draft E-ISO, (m)
draft F-ISO);
IUPAC name 3,6-dichloropyridine-2-carboxylic acid
Chemical Abstracts name 3,6-dichloro-2-pyridinecarboxylic
acid
Other names 3,6-DCP CAS RN [1702-17-6] EEC
no. 216-935-4 Development codes Dowco
290 (Dow)
clopyralid-olamine
Common name clopyralid-olamine
CAS RN [57754-85-5]
PHYSICAL CHEMISTRY
clopyralid
Mol. wt. 192.0 M.f. C6H3Cl2NO2 Form Colourless
crystals. M.p. 151-152 ºC V.p. 1.33
mPa (pure, 24 ºC); 1.36 mPa (tech., 25 ºC) KOW logP
= -1.81 (pH 5), -2.63 (pH 7), -2.55 (pH 9), 1.07 (unionised, 25 °C) S.g./density 1.57
(20 ºC) Solubility Pure (99.2%) 7.85 (in distilled
water), 118 (pH 5), 143 (pH 7), 157 (pH 9) (all in g/l, 20 ºC). In acetonitrile
121, n-hexane 6, methanol 104 (all in g/kg). Forms water-soluble salts, for
example potassium, solubility >300 g/l (25 ºC). Stability Decomposes
above m.p. Stable in acidic media and to light; on hydrolysis, DT50 >30
d at pH range 5-9 (25 ºC) in sterile water. pKa 2 F.p. No
flashing exhibited in ignition test
clopyralid-olamine
Mol. wt. 253.1 M.f. C8H10Cl2N2O3 Solubility In
water 560 g/l (25 ºC).
Biochemistry Synthetic auxin (acting like indolylacetic acid). Mode of action Selective systemic herbicide, absorbed by the leaves and roots, with translocation both acropetally and basipetally, and accumulation in meristematic tissue. Exhibits an auxin-type reaction. Acts on cell elongation and respiration.
clopyralid
Uses Post-emergence control of many annual and perennial broad-leaved weeds of the families Polygonaceae, Compositae, Leguminosae, and Umbelliferae, in sugar beet, fodder beet, oilseed rape, maize, brassicas, onions, leeks, strawberries, flax and grassland. Provides particularly good control of creeping thistle (Cirsium arvense), perennial sow-thistle, coltsfoot, mayweeds, and Polygonum spp. Phytotoxicity Good crop tolerance of graminaceous, cruciferous crops. Formulation types SL.
