Products
LINURON
Herbicide: urea
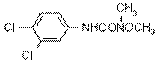
NOMENCLATURE
Common name linuron (BSI, E-ISO, (m) F-ISO,
ANSI, WSSA, JMAF)
IUPAC name 3-(3,4-dichlorophenyl)-1-methoxy-1-methylurea
Chemical Abstracts name N'-(3,4-dichlorophenyl)-N-methoxy-N-methylurea
CAS RN [330-55-2] EEC no. 206-356-5 Development
codes DPX-Z326 (Du Pont); AE F002810 (AgrEvo); Hoe 02 810
(Hoechst)
PHYSICAL CHEMISTRY
Composition Tech. is ³94% pure. Mol.
wt. 249.1 M.f. C9H10Cl2N2O2 Form Colourless
crystals. M.p. 93-95 ºC V.p.
0.051 mPa (20 ºC); 7.1 mPa (50 ºC) (EC method) KOW logP
= 3.00 Henry 2.0 ´ 10-4 Pa m3 mol-1 (20 °C) S.g./density 1.49
(20 ºC) Solubility In water 63.8 mg/l (20 ºC,
pH 7). In acetone 500, benzene, ethanol 150, xylene 130 (all in g/kg,
25 ºC). Readily soluble in dimethylformamide, chloroform, and
diethyl ether. Moderately soluble in aromatic hydrocarbons. Sparingly
soluble in aliphatic hydrocarbons. Stability Stable
at m.p. and in aqueous solution at pH 5, 7 & 9; DT50 945 d
at all three pH values.
APPLICATIONS
Biochemistry Photosynthetic electron transport inhibitor
at the photosystem II receptor site. Mode of action Selective
systemic herbicide, absorbed principally by the roots but also by the
foliage, with translocation primarily acropetally in the xylem. Uses Pre-
and post-emergence control of annual grass and broad-leaved weeds,
and some seedling perennial weeds, in asparagus, artichokes, carrots,
parsley, fennel, parsnips, herbs and spices, celery, celeriac, onions,
leeks, garlic, potatoes, peas, field beans, soya beans, cereals,maize,
sorghum, cotton, flax, sunflowers, sugar cane, ornamentals, established
vines, bananas, cassava, coffee, tea, rice, peanuts, ornamental trees
and shrubs, and other crops. Formulation types EC;
SC; WP.
