Products
METOLACHLOR
Herbicide: chloroacetamide
NOMENCLATURE
Common name metolachlor (BSI, E-ISO, ANSI,
WSSA); métolachlore ((m) F-ISO)
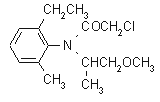
IUPAC name 2-chloro-6'-ethyl-N-(2-methoxy-1-methylethyl)aceto-o-toluidide
Chemical Abstracts name 2-chloro-N-(2-ethyl-6-methylphenyl)-N-(2-methoxy-1-methylethyl)acetamide
CAS RN [51218-45-2] EEC no. 257-060-8 Development
codes CGA 24 705 (Ciba-Geigy)
PHYSICAL CHEMISTRY
Composition Racemic mixture of (1S)- and (1R)- isomers. Mol.
wt. 283.8 M.f. C15H22ClNO2 Form Colourless
to light tan liquid. M.p. -62.1 °C B.p. 100 ºC/0.001
mmHg V.p. 4.2 mPa (25 ºC) (OECD 104) KOW logP
= 2.9 (25 ºC) Henry 2.4 × 10-3 Pa
m3 mol-1 (calc.) S.g./density 1.12 (20 ºC) Solubility In
water 488 mg/l (25 ºC) (OECD 105). Miscible with benzene, toluene,
ethanol, acetone, xylene, hexane, dimethylformamide, ethylene dichloride,
cyclohexanone, methanol, octanol, and dichloromethane. Insoluble in
ethylene glycol, propylene glycol, and petroleum ether. Stability Stable
up to c. 275 ºC. Hydrolysed by strong alkalis and strong mineral
acids. On hydrolysis in buffer (20 ºC), DT50 (calc.) >200 d
(2≤pH ≤10). F.p. 190 °C (1013 mbar)
APPLICATIONS
Biochemistry Cell division inhibitor. Maize tolerance
of chloroacetamides is attributed to rapid detoxification by glutathione
transferases. Mode of action Selective herbicide,
absorbed predominantly by the hypocotyls and shoots. Inhibits germination. Uses Control
of annual grasses and some broad-leaved weeds in maize, sorghum, cotton,
sugar beet, fodder beet, sugar cane, potatoes, peanuts, soya beans,
safflowers, sunflowers, various vegetables, fruit and nut trees, and
woody ornamentals. Applied pre-emergence, pre-plant incorporated or
early post-emergence at, 1.0-2.5 kg a.i./ha. Often used in combination
with broad-leaved herbicides, to extend the spectrum of activity. Phytotoxicity Well
tolerated by most broad-leaved crops, maize, sorghum (safened with
fluxofenim or oxabetrinil). Formulation types EC;
FW; GR; SC.
