Products
THIFENSULFURON-METHYL
Herbicide: sulfonylurea
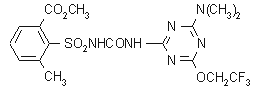
NOMENCLATURE
tribenuron-methyl
Chemical Abstracts name methyl 2-[[[[(4-methoxy-6-methyl-1,3,5-triazin-2-yl)methylamino]carbonyl]amino]sulfonyl]benzoate
CAS RN [101200-48-0] EEC no. 401-190-1 Development
codes DPX-L5300 (Du Pont); L5300
tribenuron
Common name tribenuron (BSI, ANSI, draft E-ISO)
IUPAC name 2-[4-methoxy-6-methyl-1,3,5-triazin-2-yl(methyl)carbamoylsulfamoyl]benzoic
acid
Chemical Abstracts name 2-[[[[(4-methoxy-6-methyl-1,3,5-triazin-2-yl)methylamino]carbonyl]amino]sulfonyl]benzoic
acid
CAS RN [106040-48-6]
PHYSICAL CHEMISTRY
tribenuron-methyl
Composition Tech. is >95%. Mol. wt. 395.4 M.f. C15H17N5O6S Form Light
brown, odourless solid. M.p. 141 ºC V.p.
5.2 × 10-5 mPa (25 ºC) KOW logP = -0.44 (pH 7) Henry 1.03 × 10-8
Pa m3 mol-1 S.g./density 1.5 (25 ºC) Solubility In
water 0.05 (pH 5), 2.04 (pH 7) (both in g/l, 20 ºC). In acetone 43.8,
acetonitrile 54.2, carbon tetrachloride 3.12, ethyl acetate 17.5, hexane 0.028,
and methanol 3.39 (all in mg/l, 25 °C). Stability Stable
at 45 ºC. On hydrolysis (45 ºC), stable at pH 8-10 but rapid loss
at pH <7 or pH >12. Relatively unstable in most organic solvents. pKa 5
APPLICATIONS
tribenuron-methyl
Biochemistry Branched chain amino acid synthesis (ALS or AHAS)
inhibitor. Acts by inhibiting biosynthesis of the essential amino acids valine
and isoleucine, hence stopping cell division and plant growth. Selectivity
derives from rapid metabolism in the crop. Metabolic basis of selectivity in
sulfonylureas reviewed (M. K. Koeppe & H. M. Brown, Agro-Food-Industry, 6,
9-14 (1995)). Mode of action Rapidly absorbed by foliage
and roots and translocated throughout the plant. Susceptible plants cease growth
almost immediately after post-emergence treatment and are killed in 7-21 days.
Surfactants increase the activity of tribenuron-methyl on certain broad-leaved
weeds. Uses Post-emergence control of broad-leaved weeds
in spring and winter cereals, at 9-30 g/ha. Formulation types TB;
WG.
