Products
TEBUFENPYRAD
Acaricide: pyrazole (acaricide)
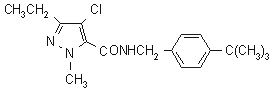
NOMENCLATURE
Common name tebufenpyrad (BSI, draft ISO)
IUPAC name N-(4-tert-butylbenzyl)-4-chloro-3-ethyl-1-methylpyrazole-5-carboxamide
Chemical Abstracts name 4-chloro-N[[4-(1,1-dimethylethyl)phenyl]methyl]-3-ethyl-1-methyl-1H-pyrazole-5-carboxamide
CAS RN [119168-77-3] Development codes AC
801757 (Cyanamid); MK-239 (Mitsubishi Kasei)
PHYSICAL CHEMISTRY
Mol. wt. 333.9 M.f. C18H24ClN3O Form Colourless
crystals. M.p. 61-62 ºC V.p. <1 ´ 10-2
mPa (25 ºC) KOW logP = 5.04 (25 ºC) Henry <1.25 ´ 10-3
Pa m3 mol-1 (calc.) S.g./density 1.0214 Solubility In
water 2.8 mg/l (25 ºC). Soluble in acetone, methanol, chloroform, acetonitrile,
hexane, and benzene. Stability Stable to hydrolysis, DT50 >28
d (pH 5, 7 and 9).
APPLICATIONS
Biochemistry Mitochondrial respiration inhibitor. Acts as an
inhibitor of the electron transport chain at Site I. Does not act as a specific
muscle or nerve poison. Mode of action Non-systemic acaricide
active by contact and ingestion. Exhibits translaminar movement following application
to leaves, and thus inhibits the development of mite eggs oviposited on the undersides
of leaves. Uses Control of all stages of Tetranychus,
Panonychus, Oligonychus, and Eotetranychus spp. on top fruit, vines, citrus,
vegetables, hops, ornamentals, melons, and cotton. Applied at 50-200 g/ha. Formulation
types EC; EW; WG; WP.
